|

|
Data Sheet
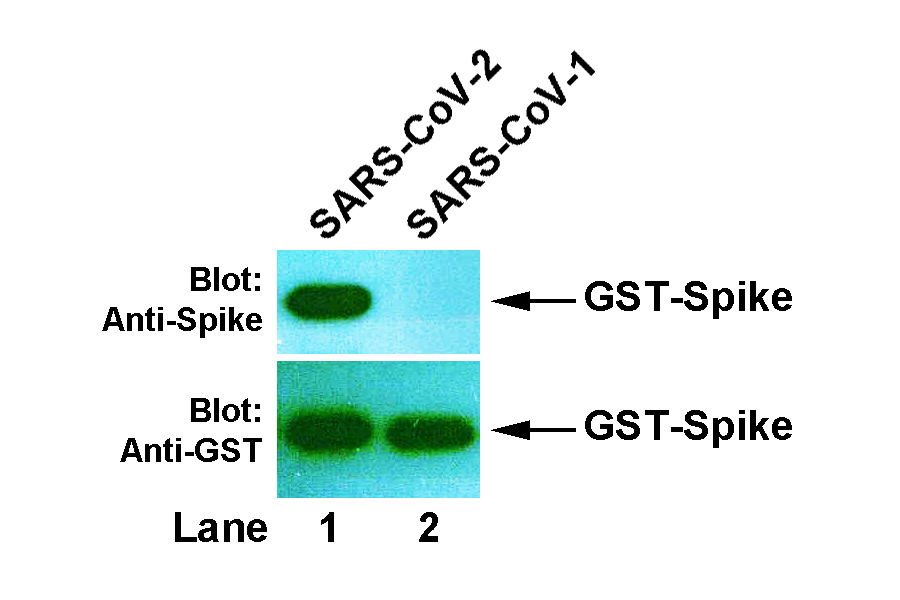
BackgroundCoronaviruses are a family of enveloped positive-sense RNA viruses ranging from 60 nm to 140 nm in diameter with trimeric spike proteins on its surface, which gives it a crown-like appearance under electron microscope. Each spike protein is a glycoprotein that comprises two functional subunits: S1 subunit is responsible for binding to the host cell receptor and S2 subunit is for the fusion of the viral and cellular membranes. Angiotensin converting enzyme 2 (ACE2) has been identified as the receptor for both SARS-CoV-1 and SARS-CoV-2, the latter causing coronavirus disease 2019 (COVID-19). The receptor-binding domain (RBD) is a subdomain of S1, which interacts with the extracellular binding site on ACE2 known as the peptidase domain (PD). Such interaction is central for virus transmission and thus considered as primary target for antiviral drug development. SourceThis is a mouse monoclonal antibody raised against the RBD fragment (N437 - Y508) of the spike protein from SARS-CoV-2. Gene SymbolSPIKE_SARS2 IsotypeIgG1 Physical FormFreeze-dried powder from 1 × PBS solution SpecificityThis antibody detects the Coronavirus spike protein of SARS-CoV-2, but not that of SARS-CoV-1. Molecular Weight141 kDa (full-length), but it appears as ~180 kDa in SDS-PAGE. ApplicationWestern blotting (WB) with dilution range: 1:1,000 - 10,000; Other applications have not been tested. StorageStore freeze-dried powder at 2 - 8°C upon arrival. When ready to use, rehydrate with 0.1 ml dH2O and centrifuge if not clear. For long-term storage, make aliquots and keep them at -20°C or below. Avoid repeated freezing and thawing cycles. Data>> Western blot: GST-Spike recombinant proteins of SARS-CoV-2 (Lane 1) and SARS-CoV-1 (Lane 2) were expressed in E. coli, then bacterial lysate was prepared and resolved in 10% SDS-PAGE. The membrane was probed with anti-Spike antibody (8E2, upper panel), strip washed and re-probed with anti-GST antibody (2E5, lower panel).
Important NoteThis product is intended for research use only, not for use in human therapeutic or diagnostic procedures. |

|
Copyright © Lanleys International Inc. All rights reserved.
|